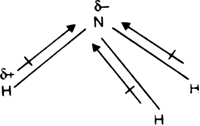
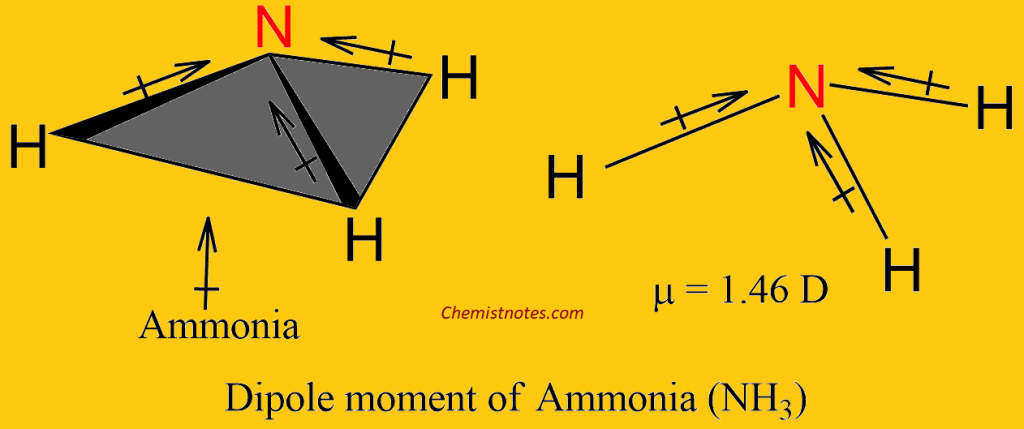
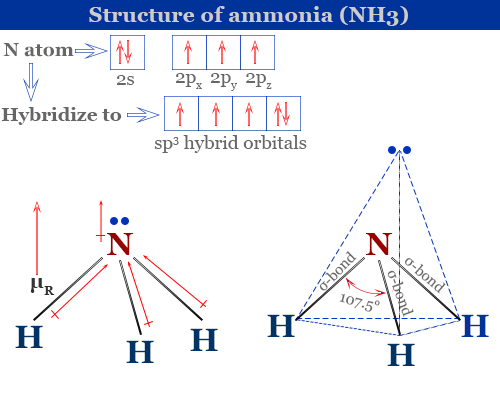
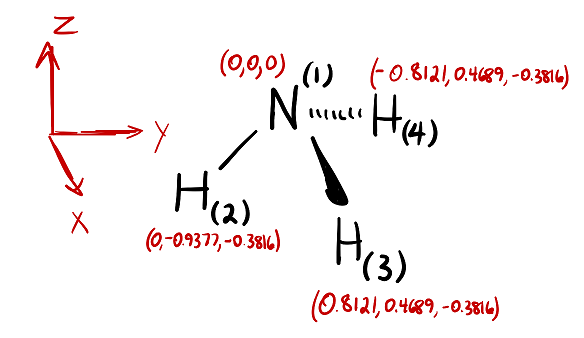
NH_3 (ammonia) is a molecule with a trigonal pyramidal shape. Is it polar? Explain. | Homework.Study.com
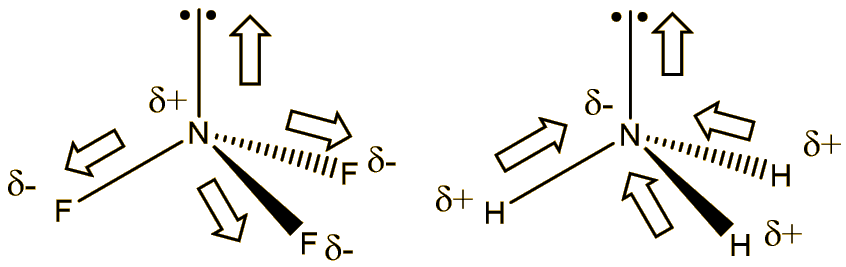
Why is the moment dipole for ammonia (1.64D substantially higher than that of ammonia trifluoride (0.24D) although the latter contains the highly electronegative flourine? - Quora

Can you explain why NH3 has such a large dipole moment compared with NF3? Show work. | Homework.Study.com
Which out of ammonia (NH3) and NF3 has higher dipole moment and why? - Sarthaks eConnect | Largest Online Education Community
Dipole moment of H2O is higher than that of NH3. Explain. - Sarthaks eConnect | Largest Online Education Community

Dipole Moment as a Possible Diagnostic Descriptor of the Conformational Isomerism of the Ammonia Molecule | Semantic Scholar