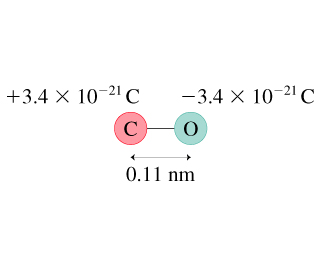Dipole moment functions of the CO molecule in the X 1 + and A 1 states.... | Download Scientific Diagram
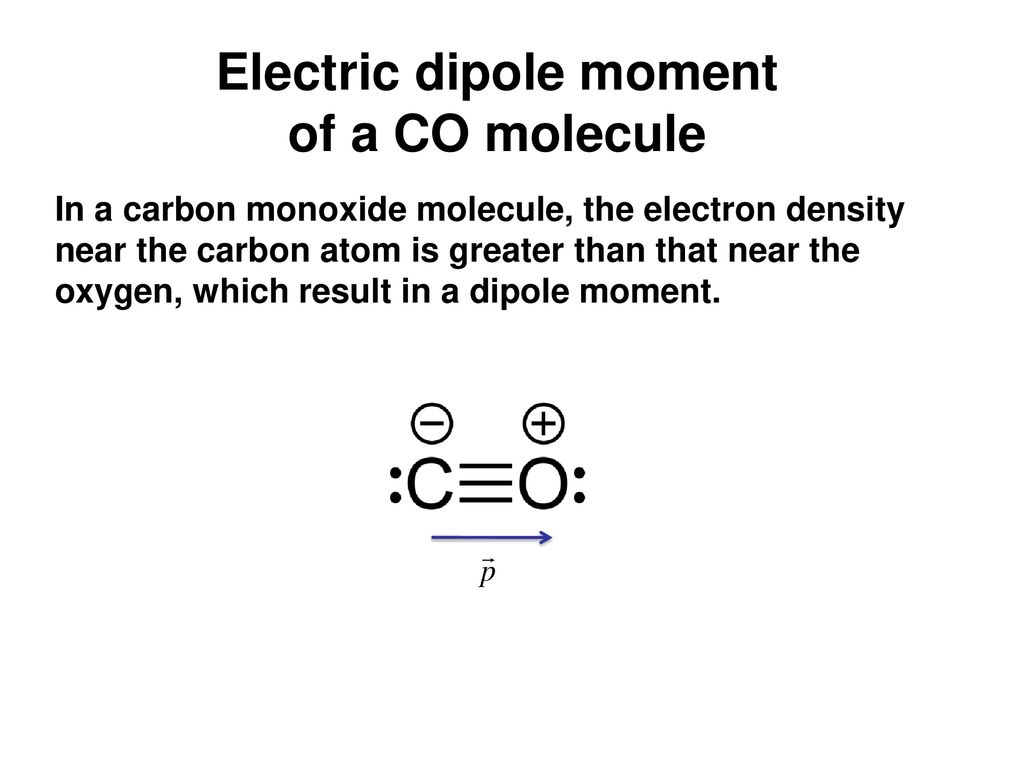
Why is the dipole moment of CO is very low? - Sarthaks eConnect | Largest Online Education Community
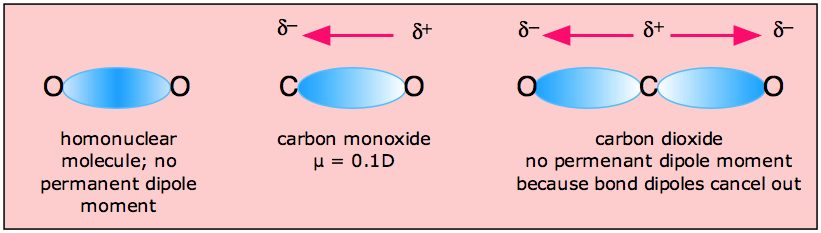
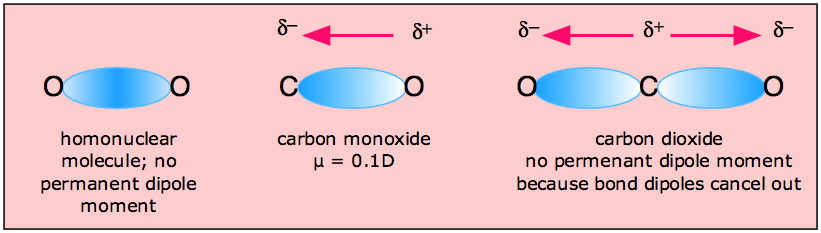
Molecules of H2O and CO have dipole moments, but CO2 does not. How can we use the sketch of the geometry of these molecules to explain this? - Quora
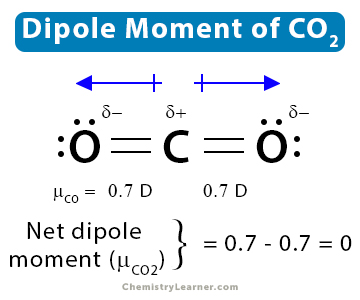
Minerals | Free Full-Text | CO2 Dipole Moment: A Simple Model and Its Implications for CO2-Rock Interactions
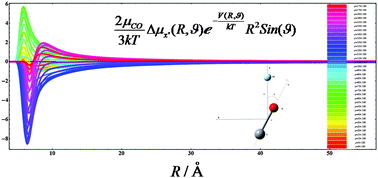
The CO–Ne van der Waals complex: ab initio intermolecular potential energy, interaction induced electric dipole moment and polarizability surfaces, and second virial coefficients - Physical Chemistry Chemical Physics (RSC Publishing)

Dipole moment of so2, h2o, ccl4, chcl3, cis & trans alkenes, co2, nh3, bf3, ch4 & organic compounds - YouTube

Correct order of dipole moment among NFZ, Co, and NH, is se answer: NF, > NH, > CO2 NH, > NF3 > CO2 NF, > CO,> NH NH, > CO2 > NF
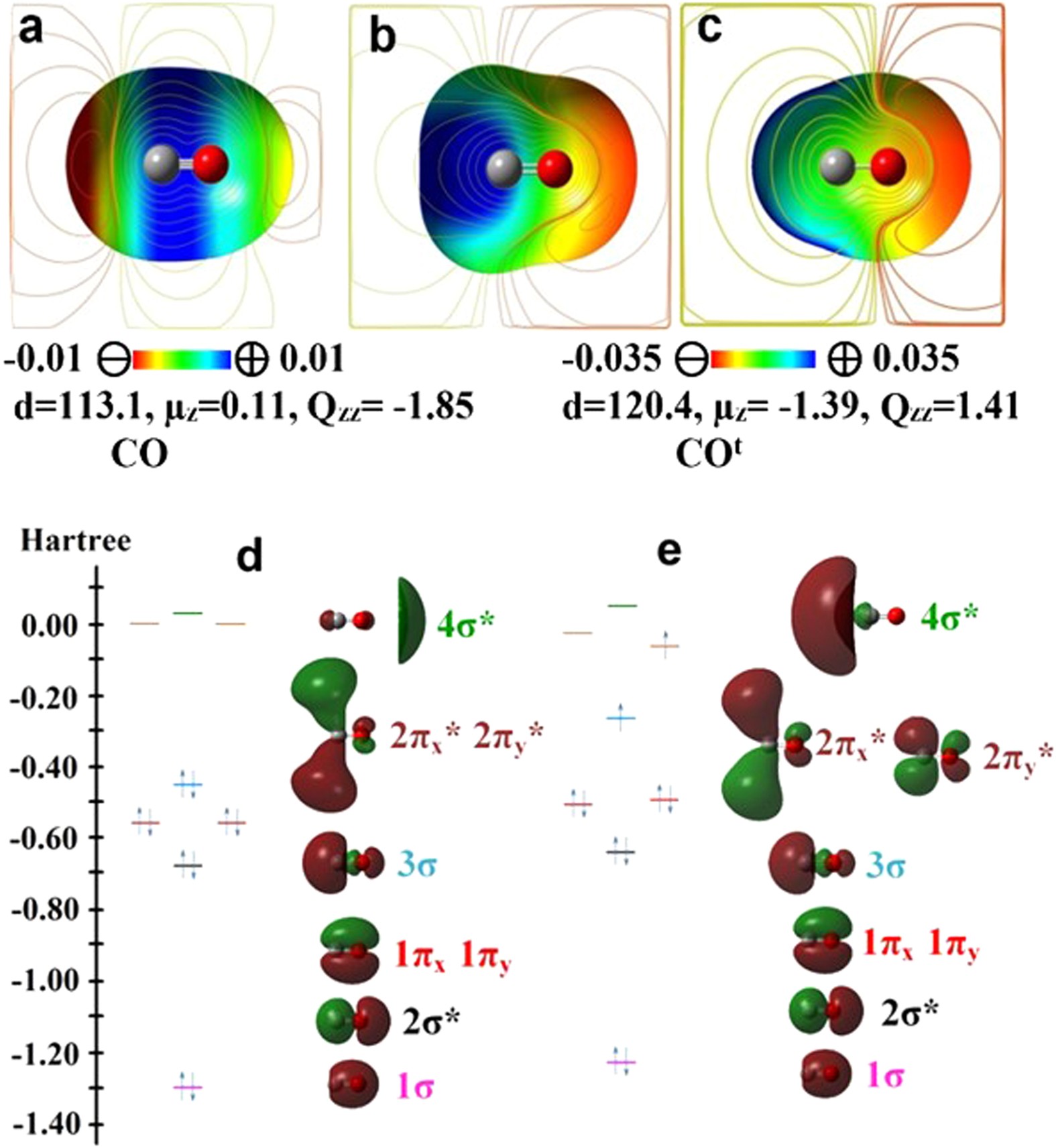
Intriguing Electrostatic Potential of CO: Negative Bond-ends and Positive Bond-cylindrical-surface | Scientific Reports
Carbon dioxide has two polar bonds, but it is a non-polar molecule. Who can explain this further? - Quora

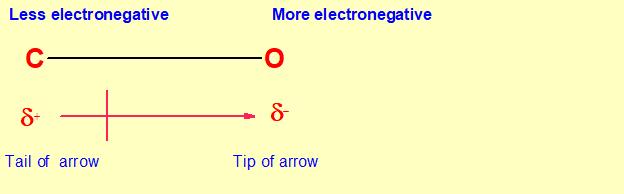
Must polar bonds give rise to polar molecules? And, why is water a polar molecule? | anhourofchemaday

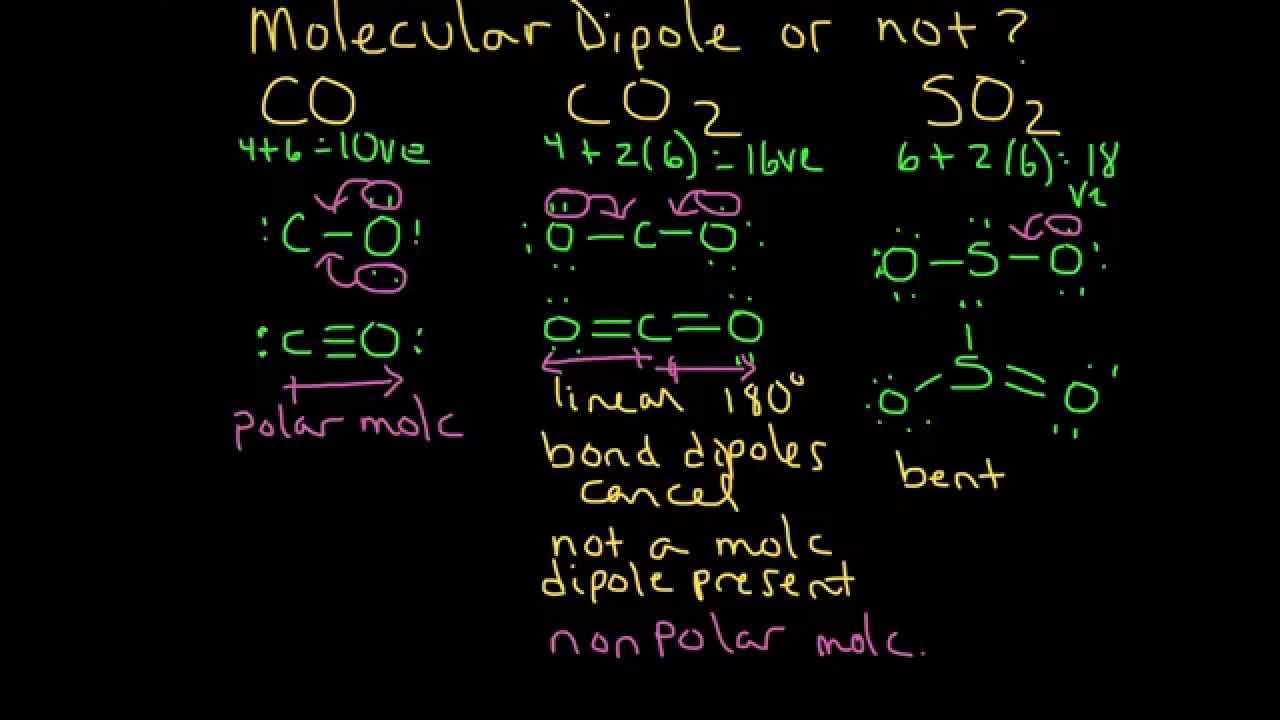





![Kannada] Which of the following possess net dipole moment? Kannada] Which of the following possess net dipole moment?](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/6848041.webp)