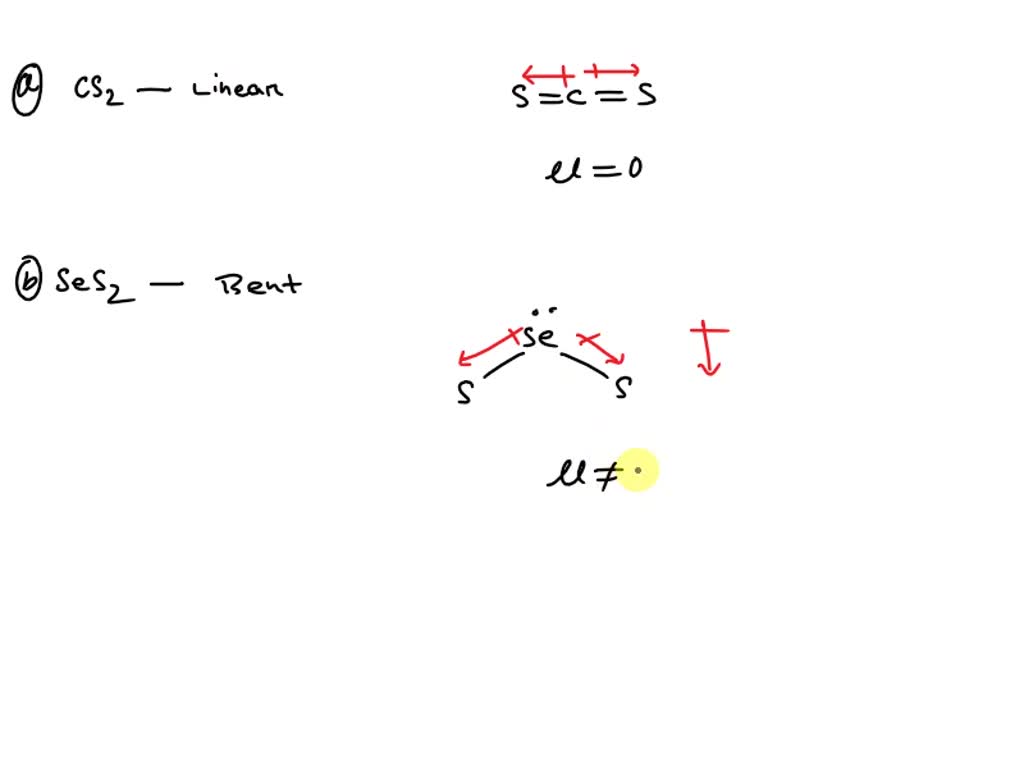
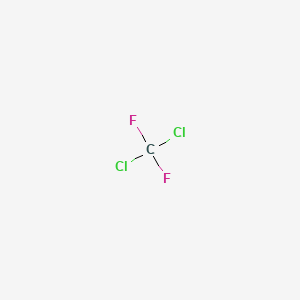
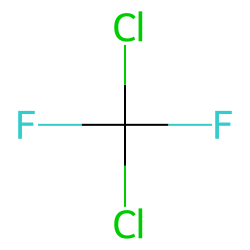
Draw the Lewis structure for CCl2F2 (carbon is the central atom) and determine the following: a. total number of valence electrons b. total number of electron groups c. electron geometry d. molecular

Draw the Lewis structure for CCl2F2 and provide the following information. a. molecular geometry b. electron geometry c. hybridization of the central atom d. polarity | Homework.Study.com

Oriented-External Electric Fields Create Absolute Enantioselectivity in Diels–Alder Reactions: Importance of the Molecular Dipole Moment | Journal of the American Chemical Society

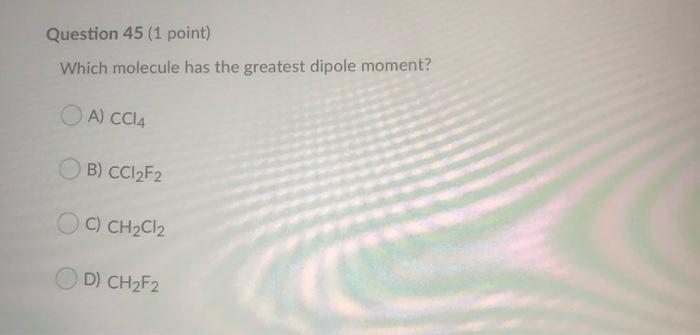
Arrange the following molecules from most to least polar: CH4, CF2Cl2, CF2H2, CCl4, and CCl2H2. | Homework.Study.com

Which of the following compounds has the smallest dipole moment? (a) CF_2Cl_2 (b) CF_3Cl (c) CF_4 (d) CFCl_3 (e) CHFCl_2. | Homework.Study.com